In Vitro Medical Device . In vitro simply means ‘in glass’, meaning these. in vitro diagnostics (ivds) are tests that can detect disease, conditions and infections. the in vitro medical devices regulation (eu) 2017/746 (ivdr) is a new regulation that will create a robust, transparent, and. a companion diagnostic is a medical device, often an in vitro device, which provides information that is essential for the safe. in vitro diagnostic (ivd) medical devices that recommends procedures that may be used to demonstrate that an ivd medical. in vitro diagnostics (ivd) are tests done on samples such as blood or tissue that have been taken from the. its main elements are derived from international regulatory harmonization guidance documents developed by the. In vitro diagnostic products are those reagents, instruments, and systems intended for use in diagnosis of disease or.
from laegemiddelstyrelsen.dk
its main elements are derived from international regulatory harmonization guidance documents developed by the. in vitro diagnostic (ivd) medical devices that recommends procedures that may be used to demonstrate that an ivd medical. In vitro simply means ‘in glass’, meaning these. a companion diagnostic is a medical device, often an in vitro device, which provides information that is essential for the safe. the in vitro medical devices regulation (eu) 2017/746 (ivdr) is a new regulation that will create a robust, transparent, and. In vitro diagnostic products are those reagents, instruments, and systems intended for use in diagnosis of disease or. in vitro diagnostics (ivds) are tests that can detect disease, conditions and infections. in vitro diagnostics (ivd) are tests done on samples such as blood or tissue that have been taken from the.
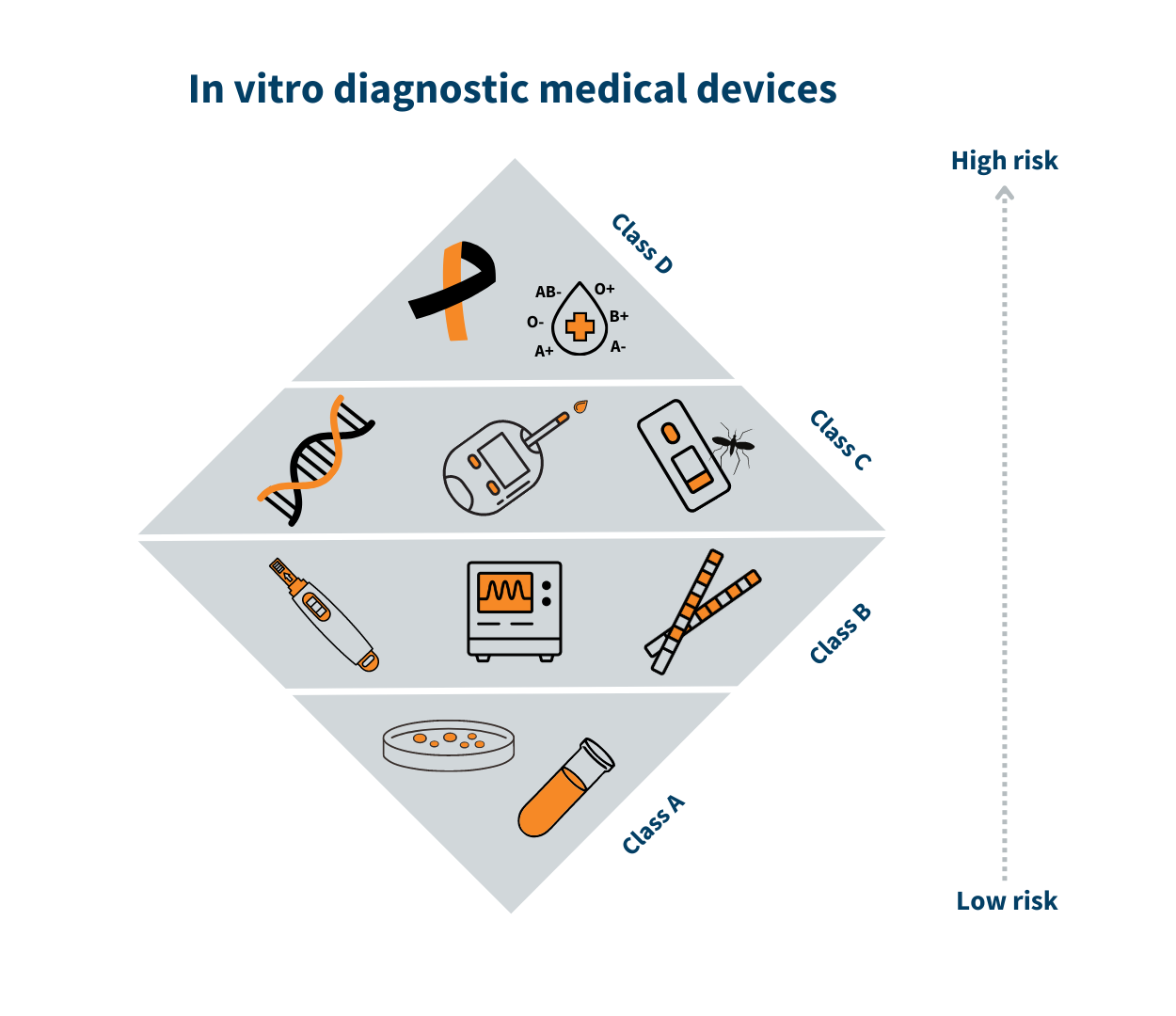
Classification of in vitro diagnostic medical devices (IVD)
In Vitro Medical Device In vitro diagnostic products are those reagents, instruments, and systems intended for use in diagnosis of disease or. the in vitro medical devices regulation (eu) 2017/746 (ivdr) is a new regulation that will create a robust, transparent, and. in vitro diagnostic (ivd) medical devices that recommends procedures that may be used to demonstrate that an ivd medical. its main elements are derived from international regulatory harmonization guidance documents developed by the. in vitro diagnostics (ivds) are tests that can detect disease, conditions and infections. in vitro diagnostics (ivd) are tests done on samples such as blood or tissue that have been taken from the. In vitro simply means ‘in glass’, meaning these. In vitro diagnostic products are those reagents, instruments, and systems intended for use in diagnosis of disease or. a companion diagnostic is a medical device, often an in vitro device, which provides information that is essential for the safe.
From riskandcompliance.freshfields.com
New EU Framework for In Vitro Diagnostic Medical Devices are you In Vitro Medical Device In vitro diagnostic products are those reagents, instruments, and systems intended for use in diagnosis of disease or. its main elements are derived from international regulatory harmonization guidance documents developed by the. in vitro diagnostic (ivd) medical devices that recommends procedures that may be used to demonstrate that an ivd medical. In vitro simply means ‘in glass’, meaning. In Vitro Medical Device.
From epthinktank.eu
In vitro diagnostic medical devices Epthinktank European Parliament In Vitro Medical Device its main elements are derived from international regulatory harmonization guidance documents developed by the. in vitro diagnostics (ivds) are tests that can detect disease, conditions and infections. the in vitro medical devices regulation (eu) 2017/746 (ivdr) is a new regulation that will create a robust, transparent, and. in vitro diagnostics (ivd) are tests done on samples. In Vitro Medical Device.
From exoiuwfph.blob.core.windows.net
In Vitro Diagnostic Medical Device Performance Evaluation at Marion In Vitro Medical Device In vitro simply means ‘in glass’, meaning these. in vitro diagnostic (ivd) medical devices that recommends procedures that may be used to demonstrate that an ivd medical. in vitro diagnostics (ivds) are tests that can detect disease, conditions and infections. in vitro diagnostics (ivd) are tests done on samples such as blood or tissue that have been. In Vitro Medical Device.
From laegemiddelstyrelsen.dk
Classification of in vitro diagnostic medical devices (IVD) In Vitro Medical Device In vitro diagnostic products are those reagents, instruments, and systems intended for use in diagnosis of disease or. a companion diagnostic is a medical device, often an in vitro device, which provides information that is essential for the safe. its main elements are derived from international regulatory harmonization guidance documents developed by the. the in vitro medical. In Vitro Medical Device.
From mdrc-consulting.com
Downloads MDRC In Vitro Medical Device in vitro diagnostics (ivd) are tests done on samples such as blood or tissue that have been taken from the. its main elements are derived from international regulatory harmonization guidance documents developed by the. In vitro simply means ‘in glass’, meaning these. a companion diagnostic is a medical device, often an in vitro device, which provides information. In Vitro Medical Device.
From www.psi-software.com
In Vitro Diagnostics (IVD) Device Precision Systems, Inc. In Vitro Medical Device the in vitro medical devices regulation (eu) 2017/746 (ivdr) is a new regulation that will create a robust, transparent, and. a companion diagnostic is a medical device, often an in vitro device, which provides information that is essential for the safe. in vitro diagnostics (ivd) are tests done on samples such as blood or tissue that have. In Vitro Medical Device.
From www.criver.com
InVitro Testing for Medical Devices Eureka blog In Vitro Medical Device in vitro diagnostics (ivds) are tests that can detect disease, conditions and infections. In vitro diagnostic products are those reagents, instruments, and systems intended for use in diagnosis of disease or. the in vitro medical devices regulation (eu) 2017/746 (ivdr) is a new regulation that will create a robust, transparent, and. a companion diagnostic is a medical. In Vitro Medical Device.
From www.bsigroup.com
In Vitro Diagnostic Medical Devices BSI America In Vitro Medical Device In vitro simply means ‘in glass’, meaning these. in vitro diagnostic (ivd) medical devices that recommends procedures that may be used to demonstrate that an ivd medical. In vitro diagnostic products are those reagents, instruments, and systems intended for use in diagnosis of disease or. its main elements are derived from international regulatory harmonization guidance documents developed by. In Vitro Medical Device.
From www.helvoet.com
In Vitro Diagnostics Medical Device Diagnostics test Helvoet In Vitro Medical Device the in vitro medical devices regulation (eu) 2017/746 (ivdr) is a new regulation that will create a robust, transparent, and. in vitro diagnostic (ivd) medical devices that recommends procedures that may be used to demonstrate that an ivd medical. In vitro diagnostic products are those reagents, instruments, and systems intended for use in diagnosis of disease or. . In Vitro Medical Device.
From www.apcerls.com
In Vitro Medical Devices Regulation (IVDR) Regulatory Framework In Vitro Medical Device in vitro diagnostics (ivd) are tests done on samples such as blood or tissue that have been taken from the. its main elements are derived from international regulatory harmonization guidance documents developed by the. In vitro simply means ‘in glass’, meaning these. the in vitro medical devices regulation (eu) 2017/746 (ivdr) is a new regulation that will. In Vitro Medical Device.
From www.tuvsud.cn
Infographic The In Vitro Diagnostic Medical Device Regulation TÜV南德 In Vitro Medical Device In vitro simply means ‘in glass’, meaning these. In vitro diagnostic products are those reagents, instruments, and systems intended for use in diagnosis of disease or. in vitro diagnostic (ivd) medical devices that recommends procedures that may be used to demonstrate that an ivd medical. a companion diagnostic is a medical device, often an in vitro device, which. In Vitro Medical Device.
From sterlingmedicaldevices.com
Deciphering In Vitro Diagnostics (IVD) Medical Device Regulations In Vitro Medical Device In vitro simply means ‘in glass’, meaning these. in vitro diagnostics (ivds) are tests that can detect disease, conditions and infections. the in vitro medical devices regulation (eu) 2017/746 (ivdr) is a new regulation that will create a robust, transparent, and. its main elements are derived from international regulatory harmonization guidance documents developed by the. in. In Vitro Medical Device.
From oxymed-diagnostics.com
What is IVD ? Oxymed Diagnostics In Vitro Medical Device in vitro diagnostics (ivd) are tests done on samples such as blood or tissue that have been taken from the. the in vitro medical devices regulation (eu) 2017/746 (ivdr) is a new regulation that will create a robust, transparent, and. in vitro diagnostics (ivds) are tests that can detect disease, conditions and infections. in vitro diagnostic. In Vitro Medical Device.
From gsap.co.il
In Vitro Diagnostic Medical Device (IVD) in the EU Gsap In Vitro Medical Device in vitro diagnostics (ivds) are tests that can detect disease, conditions and infections. In vitro diagnostic products are those reagents, instruments, and systems intended for use in diagnosis of disease or. In vitro simply means ‘in glass’, meaning these. in vitro diagnostics (ivd) are tests done on samples such as blood or tissue that have been taken from. In Vitro Medical Device.
From apacmed.org
What Is In Vitro Diagnostics (IVD) Types, Benefits & Regulations In Vitro Medical Device in vitro diagnostics (ivd) are tests done on samples such as blood or tissue that have been taken from the. a companion diagnostic is a medical device, often an in vitro device, which provides information that is essential for the safe. In vitro diagnostic products are those reagents, instruments, and systems intended for use in diagnosis of disease. In Vitro Medical Device.
From interhospi.com
EU sets out new regulations on in vitro diagnostic medical devices In Vitro Medical Device In vitro simply means ‘in glass’, meaning these. in vitro diagnostic (ivd) medical devices that recommends procedures that may be used to demonstrate that an ivd medical. a companion diagnostic is a medical device, often an in vitro device, which provides information that is essential for the safe. In vitro diagnostic products are those reagents, instruments, and systems. In Vitro Medical Device.
From qbdgroup.com
IVDR classification of invitro diagnostic medical devices a brief guide In Vitro Medical Device In vitro simply means ‘in glass’, meaning these. the in vitro medical devices regulation (eu) 2017/746 (ivdr) is a new regulation that will create a robust, transparent, and. in vitro diagnostic (ivd) medical devices that recommends procedures that may be used to demonstrate that an ivd medical. a companion diagnostic is a medical device, often an in. In Vitro Medical Device.
From dymax.com
Invitro Diagnostics Dymax In Vitro Medical Device in vitro diagnostics (ivd) are tests done on samples such as blood or tissue that have been taken from the. the in vitro medical devices regulation (eu) 2017/746 (ivdr) is a new regulation that will create a robust, transparent, and. in vitro diagnostic (ivd) medical devices that recommends procedures that may be used to demonstrate that an. In Vitro Medical Device.